Yellow Pill With D 855
9 Topics Found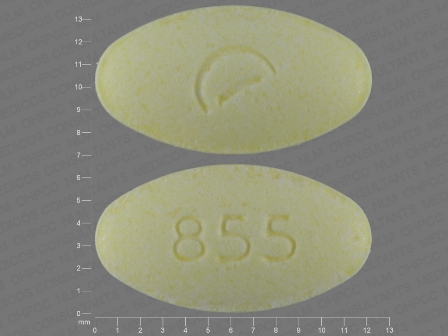
- NDC: *
- 0228-2855
- Imprint:
- 855
- Color:
- Yellow
- Shape:
- Oval
- Size:
- 12 mm
- Segments:
- 1
- Drug:
- Guanfacine
- Labeler: *
- Actavis Pharma, Inc.
- Category:
- Blood Pressure
Source: US National Library of Medicine (NLM)
Please help me figure out what this is asap. Its yellow kinda oval.855 on one side and half moon like thing on the other side.please help me asap ## Hello, Msp! How are you? This tablet is manufactured by Actavis Pharma and they list it as containing 4mgs of Guanfacine in an extended release formulation. Guanfacine is approved by the FDA to treat ADHD and hypertension. Typical side effects are listed as possibly including nausea, dizziness, somnolence, headache, constipation and abdominal pain. Is there anything else I can help with? ## What is this pill used for and is it a narcotic
yellow says watson 853 suppost to be narco need full information on medication ## I have a bottle of pills I take to work but only 3or4 of each. Now that I haven't worked I Can't identify all the pills. ## The pill which you are describing is Acetaminophen + Hydrocodone bitartrate (325 mg - 10 mg). Acetaminophen + Hydrocodone is a combination of two types of pain medicine to treat moderate to severe pain. For more information about this drug, please refer to the link below... If you have any more questions or concerns, please post back and I will be happy to help you. ## Is Watson 855. For Narco 10 mg a good Brand pill ?
Naproxen 500 mg Oral Tablet by Cipla USA Inc. ## Markings: IGand342 ## Shape: Capsule ## Color: Yellow ## Package Codes: 69097-855-07, 69097-855-12
Terazosin Hydrochloride 2 mg Oral Capsule by Apnar Pharma Lp ## Markings: AP782 ## Shape: Capsule ## Color: Yellow ## Package Codes: 24689-855-01, 24689-855-10 ## Active Ingredients: Terazosin Hydrochloride
Guanfacine 4 mg Oral Tablet, Extended Release by Actavis Pharma, Inc. ## Markings: 855 ## Shape: Oval ## Color: Yellow ## Package Codes: 0228-2855-11, 0228-2855-50 ## Active Ingredients: Guanfacine Hydrochloride
Methotrexate 2.5 mg (As Methotrexate Sodium) Oral Tablet by Roxane Laboratories, Inc ## Markings: 54323 ## Shape: Round ## Color: Yellow ## Package Codes: 0054-8550-25 ## Active Ingredients: Methotrexate Sodium
Naproxen 500 mg Oral Tablet by Clinical Solutions Wholesale, LLC ## Markings: IGand342 ## Shape: Capsule ## Color: Yellow ## Package Codes: 58118-0855-8
Guanfacine 4 mg Oral Tablet, Extended Release by Avkare, Inc. ## Markings: 855 ## Shape: Oval ## Color: Yellow ## Package Codes: 42291-327-01 ## Active Ingredients: Guanfacine Hydrochloride