Ondansetron - NDC Database (Page 14)
Ondansetron Hydrochloride 8 mg Oral Tablet, Film Coated by Bryant Ranch Prepack

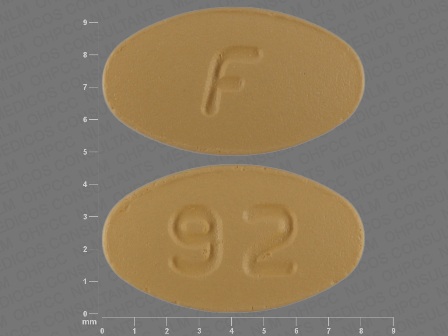
Ondansetron Hydrochloride 8 mg Oral Tablet, Film Coated by Aphena Pharma Solutions - Tennessee, LLC

Ondansetron Hydrochloride 4 mg Oral Tablet, Film Coated by Pd-rx Pharmaceuticals, Inc.
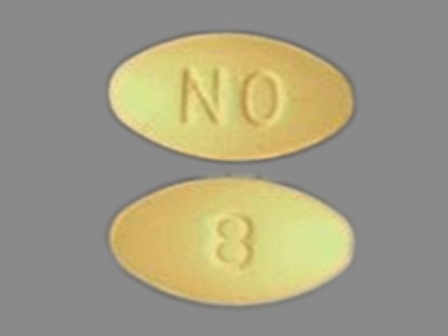
Ondansetron 8 mg (As Ondansetron Hydrochloride Dihydrate 10 mg) Oral Tablet by Life Line Home Care Services, Inc.
0069-0700 May 01, 2009
Ondansetron Hydrochloride and Dextrose 32 mg/50ml Intravenous Injection, Solution by Pfizer Labs, Division of Pfizer Inc.
Ondansetron Hydrochloride and Dextrose 32 mg/50ml Intravenous Injection, Solution by Pfizer Labs, Division of Pfizer Inc.
0069-1340 May 10, 2011
Ondansetron Hydrochloride 2 mg/ml Intramuscular; Intravenous Injection, Solution by Pfizer Laboratories Div Pfizer Inc
Ondansetron Hydrochloride 2 mg/ml Intramuscular; Intravenous Injection, Solution by Pfizer Laboratories Div Pfizer Inc
0069-1441 May 04, 2013
Ondansetron 2 mg/ml Intramuscular; Intravenous Injection by Pfizer Laboratories Div Pfizer Inc
Ondansetron 2 mg/ml Intramuscular; Intravenous Injection by Pfizer Laboratories Div Pfizer Inc
0078-0679 Aug 23, 2017
Zofran 4 mg Oral Tablet, Orally Disintegrating by Novartis Pharmaceuticals Corporation
Zofran 4 mg Oral Tablet, Orally Disintegrating by Novartis Pharmaceuticals Corporation
0078-0680 Aug 23, 2017
Zofran 8 mg Oral Tablet, Orally Disintegrating by Novartis Pharmaceuticals Corporation
Zofran 8 mg Oral Tablet, Orally Disintegrating by Novartis Pharmaceuticals Corporation
0143-2424 Jan 01, 2008
Ondansetron 24 mg (Ondansetron Hydrochloride Dihydrate 30 mg) Oral Tablet by West-ward Pharmaceutical Corp
Ondansetron 24 mg (Ondansetron Hydrochloride Dihydrate 30 mg) Oral Tablet by West-ward Pharmaceutical Corp
0143-9771 Aug 04, 2009
Ondansetron Hydrochloride and Dextrose 32 mg/50ml Intravenous Injection, Solution by West-ward Pharmaceutical Corp
Ondansetron Hydrochloride and Dextrose 32 mg/50ml Intravenous Injection, Solution by West-ward Pharmaceutical Corp
0143-9891 Jan 01, 2007
Ondansetron 4 mg/2ml Intramuscular; Intravenous Injection by West-ward Pharmaceutical Corp
Ondansetron 4 mg/2ml Intramuscular; Intravenous Injection by West-ward Pharmaceutical Corp
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.

