Lithium Carbonate - NDC Database
156 records found
Page: 1 Start a Discussion
0054-0020 Jan 13, 2004
Lico3 450 mg Extended Release Tablet by Roxane Laboratories, Inc
0054-0021 Oct 28, 2004
Lico3 300 mg Extended Release Tablet by Roxane Laboratories, Inc
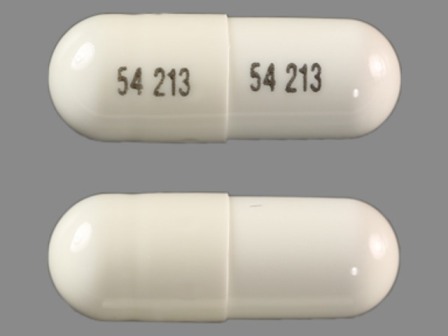
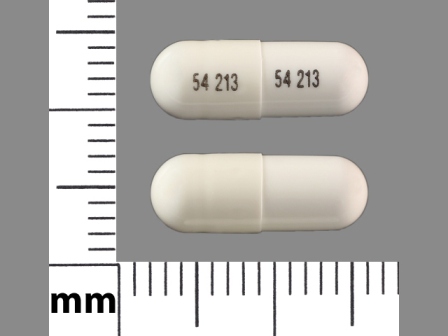
0054-2526 Jan 28, 1987
Lico3 150 mg Oral Capsule by Roxane Laboratories, Inc
0054-2527 Nov 26, 1980
Lico3 300 mg Oral Capsule by Roxane Laboratories, Inc
0054-2531 Jan 28, 1987
Lico3 600 mg Oral Capsule by Roxane Laboratories, Inc
0054-4527 Jan 29, 1982
Lico3 300 mg Oral Tablet by Roxane Laboratories, Inc
0054-8526 Jan 28, 1987
Lico3 150 mg Oral Capsule by Roxane Laboratories, Inc
0054-8527 Jul 28, 1987
Lico3 300 mg Oral Capsule by Roxane Laboratories, Inc
0054-8528 Jun 14, 1982
Lico3 300 mg Oral Tablet by Roxane Laboratories, Inc
0054-8531 Jan 28, 1987
Lico3 600 mg Oral Capsule by Roxane Laboratories, Inc
0143-1277 Sep 06, 2002
Lico3 450 mg Extended Release Tablet by West-ward Pharmaceutical Corp
0143-1300 Dec 28, 2010
Lico3 300 mg Oral Tablet by West-ward Pharmaceutical Corp
0143-3188 Mar 01, 2003
Lico3 150 mg Oral Capsule by West-ward Pharmaceutical Corp
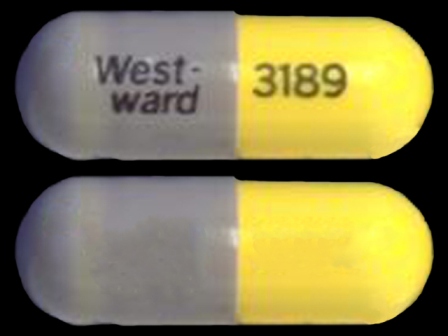
0143-3189 Mar 01, 2003
Lico3 300 mg Oral Capsule by West-ward Pharmaceutical Corp
0378-1300 Jul 25, 2012
Lico3 300 mg Extended Release Tablet by Mylan Pharmaceuticals Inc.
0904-6205 Feb 24, 2003
Lico3 150 mg Oral Capsule by Major Pharmaceuticals
31722-544 Dec 15, 2009
Lico3 150 mg Oral Capsule by Camber Pharmaceuticals Inc.
31722-545 Dec 15, 2009
Lico3 300 mg Oral Capsule by Camber Pharmaceuticals Inc.
60429-267 Jun 17, 2003
Lico3 450 mg Extended Release Tablet by Golden State Medical Supply, Inc.
60505-2504 Nov 29, 2004
Lico3 300 mg Oral Capsule by Apotex Corp
Page: 1 Start a Discussion
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us .