Exforge - NDC Database
9 records found
Page:
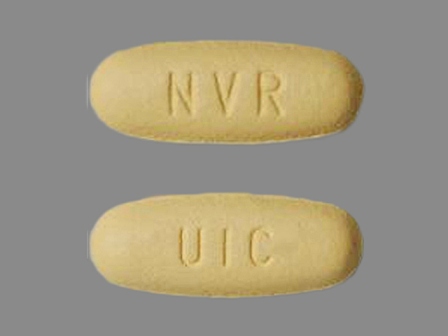
Exforge 5/160 (Amlodipine / Valsartan) Oral Tablet by Novartis Pharmaceuticals Corporation
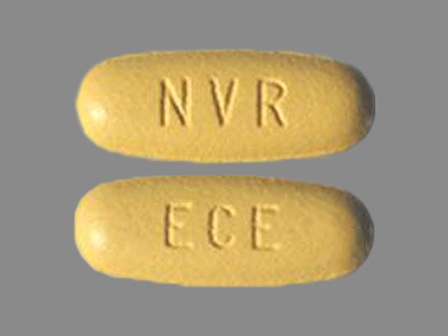
Exforge 5/320 (Amlodipine / Valsartan) Oral Tablet by Novartis Pharmaceuticals Corporation
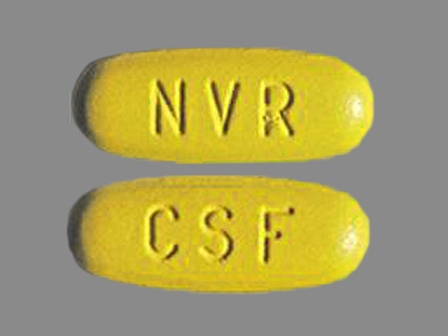
Exforge 10/320 (Amlodipine / Valsartan) Oral Tablet by Novartis Pharmaceuticals Corporation

Exforge 10/320 (Amlodipine / Valsartan) Oral Tablet by Physicians Total Care, Inc.

Exforge 5/320 (Amlodipine / Valsartan) Oral Tablet by Physicians Total Care, Inc.

Exforge 5/160 (Amlodipine / Valsartan) Oral Tablet by Physicians Total Care, Inc.
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.