Diclofenac + Misoprostol - NDC Database

Diclofenac Sodium (Enteric Coated Core) 50 mg / Misoprostol (Non-enteric Coated Mantle) 200 Mcg Oral Tablet by Watson Laboratories, Inc.

Diclofenac Sodium (Enteric Coated Core) 75 mg / Misoprostol (Non-enteric Coated Mantle) 200 Mcg Oral Tablet by Watson Laboratories, Inc.

Diclofenac Sodium (Enteric Coated Core) 50 mg / Misoprostol (Non-enteric Coated Mantle) 200 Mcg Oral Tablet by Avkare, Inc.

Diclofenac Sodium (Enteric Coated Core) 75 mg / Misoprostol (Non-enteric Coated Mantle) 200 Mcg Oral Tablet by Avkare, Inc.

Diclofenac Sodium and Misoprostol Oral Tablet, Delayed Release by Eagle Pharmaceuticals, Inc.
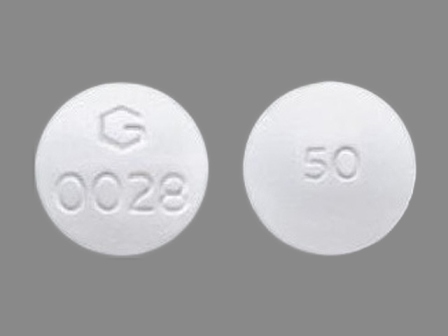
Diclofenac Sodium (Enteric Coated Core) 50 mg / Misoprostol (Non-enteric Coated Mantle) 200 Mcg Oral Tablet by Greenstone LLC
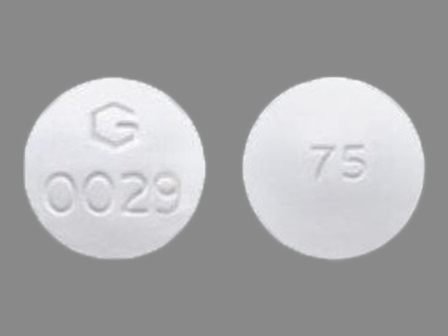
Diclofenac Sodium (Enteric Coated Core) 75 mg / Misoprostol (Non-enteric Coated Mantle) 200 Mcg Oral Tablet by Greenstone LLC

Diclofenac Sodium and Misoprostol Oral Tablet, Delayed Release by Blenheim Pharmacal, Inc.

Diclofenac Sodium and Misoprostol (Diclofenac Sodium 75 mg / Misoprostol 200 Ug) by Bryant Ranch Prepack

Diclofenac Sodium and Misoprostol Oral Tablet, Delayed Release by Bluepoint Laboratories

Diclofenac Sodium and Misoprostol Oral Tablet, Delayed Release by Bluepoint Laboratories

Diclofenac Sodium and Misoprostol Oral Tablet, Delayed Release by American Health Packaging

Diclofenac Sodium and Misoprostol Oral Tablet, Delayed Release by Preferred Pharmaceuticals, Inc.

Diclofenac Sodium and Misoprostol Oral Tablet, Delayed Release by Dash Pharmaceuticals LLC
10544-949 Oct 07, 2015
Diclofenac Sodium and Misoprostol Oral Tablet, Delayed Release by Blenheim Pharmacal, Inc.
Diclofenac Sodium and Misoprostol Oral Tablet, Delayed Release by Blenheim Pharmacal, Inc.
42367-110 Jul 15, 2014
Diclofenac Sodium and Misoprostol Oral Tablet, Delayed Release by Eagle Pharmaceuticals, Inc.
Diclofenac Sodium and Misoprostol Oral Tablet, Delayed Release by Eagle Pharmaceuticals, Inc.
50090-4618 Dec 01, 2016
Diclofenac Sodium and Misoprostol Oral Tablet, Delayed Release by A-s Medication Solutions
Diclofenac Sodium and Misoprostol Oral Tablet, Delayed Release by A-s Medication Solutions
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.