Acyclovir - NDC Database (Page 12)
0143-9657 Feb 29, 2016
Acyclovir 500 mg Intravenous Injection, Powder, Lyophilized, for Solution by West-ward Pharmaceuticals Corp
Acyclovir 500 mg Intravenous Injection, Powder, Lyophilized, for Solution by West-ward Pharmaceuticals Corp
0143-9658 Feb 29, 2016
Acyclovir 1 g/1 Intravenous Injection, Powder, Lyophilized, for Solution by West-ward Pharmaceuticals Corp
Acyclovir 1 g/1 Intravenous Injection, Powder, Lyophilized, for Solution by West-ward Pharmaceuticals Corp
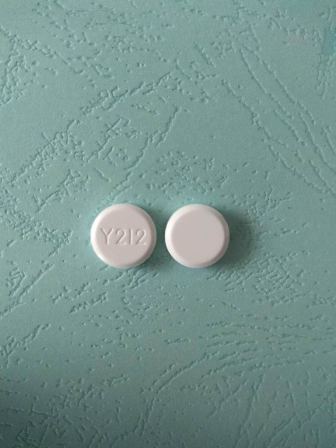
0168-0825 May 11, 2016
Acyclovir 50 mg/g Topical Ointment by E. Fougera & Co. a Division of Fougera Pharmaceuticals Inc.
Acyclovir 50 mg/g Topical Ointment by E. Fougera & Co. a Division of Fougera Pharmaceuticals Inc.
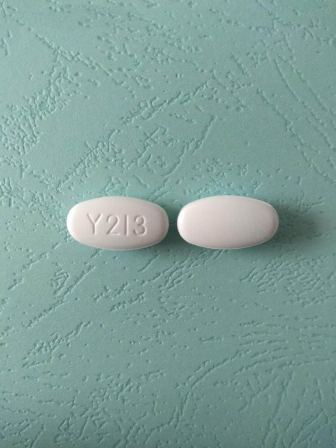
0378-4275 May 24, 2010
Valacyclovir (As Valacyclovir Hydrochloride) 500 mg Oral Tablet by Mylan Pharmaceuticals Inc.
Valacyclovir (As Valacyclovir Hydrochloride) 500 mg Oral Tablet by Mylan Pharmaceuticals Inc.
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.