Zerit
Active Ingredient(s): Stavudine, d4TFDA Approved: * June 24, 1994
Pharm Company: * BRISTOL MYERS SQUIBB
Category: HIV / AIDS
Stavudine (d4T), sold under the brand name Zerit among others, is an antiretroviral medication used to prevent and treat HIV/AIDS.[3] It is generally recommended for use with other antiretrovirals.[3] It may be used for prevention after a needlestick injury or other potential exposure.[3] However, it is not a first-line treatment.[3] It is given by mouth.[3] Common side effects include headache, diarrhea, vomiting... [wikipedia]
* May have multiple approval dates, manufacturers, or labelers.Dosage List
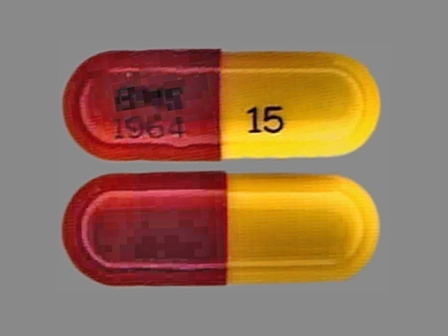
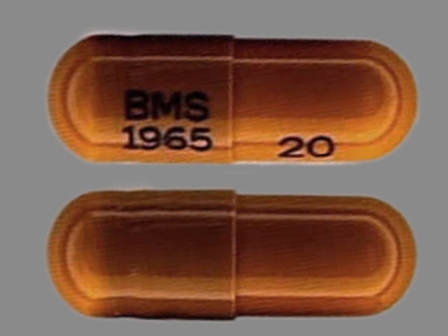
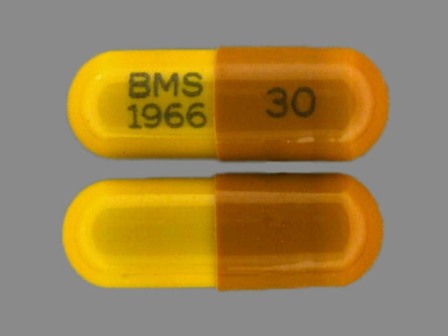
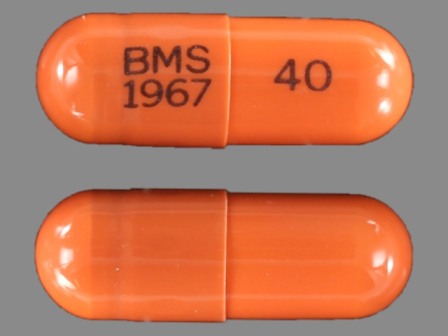



Related Brands
Drugs with the same active ingredientsPopular Topics
zeritan
hiv...