Razadyne
Active Ingredient(s): GalantamineFDA Approved: * February 28, 2001
Pharm Company: * JANSSEN PHARMA
Category: Alzheimers / Dementia
Galantamine[3] is used for the treatment of cognitive decline in mild to moderate Alzheimer's disease and various other memory impairments.[4][5] It is an alkaloid that has been isolated from the bulbs and flowers of Galanthus nivalis (Common snowdrop), Galanthus caucasicus (Caucasian snowdrop), Galanthus woronowii (Voronov's snowdrop), and some other members of the family Amaryllidaceae, such as Narcissus (daffodil), Leucojum aestivum (snowflak... [wikipedia]
* May have multiple approval dates, manufacturers, or labelers.1 Discussion
Dosage List

24 Hr Razadyne ER 8 mg Extended Release Capsule
NDC: 50458-387
Labeler:
Janssen Pharmaceuticals, Inc.

24 Hr Razadyne ER 16 mg Extended Release Capsule
NDC: 50458-388
Labeler:
Janssen Pharmaceuticals, Inc.
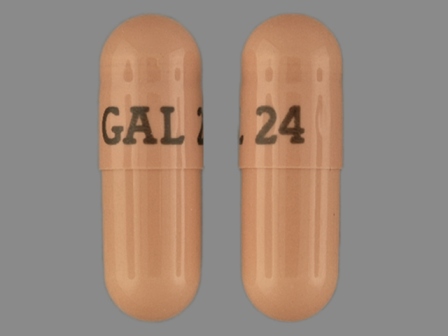
Razadyne ER 24 mg 24hr Extended Release Capsule
NDC: 50458-389
Labeler:
Janssen Pharmaceuticals, Inc.
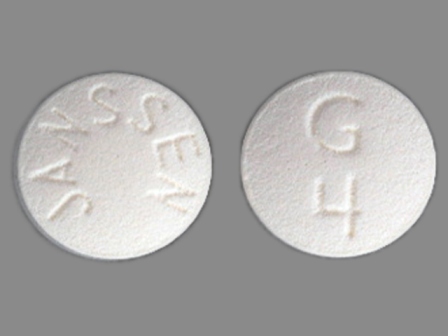
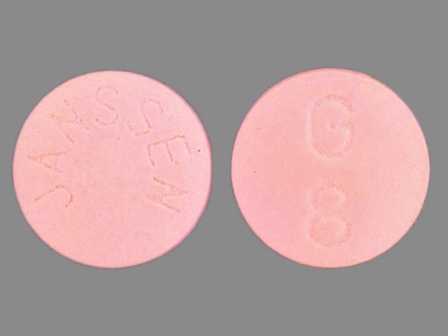

Razadyne 12 mg (Galantamine Hydrobromide 15.379 mg) Oral Tablet
NDC: 50458-398
Labeler:
Janssen Pharmaceuticals, Inc.

Razadyne 12 mg (Galantamine Hydrobromide 15.379 mg) Oral Tablet
NDC: 21695-591
Labeler:
Rebel Distributors Corp
Related Brands
Drugs with the same active ingredientsPopular Topics
razadyne IR
40mg tabs...