Onfi
Active Ingredient(s): ClobazamFDA Approved: * October 21, 2011
Pharm Company: * LUNDBECK INC
Category: Anticonvulsant
Clobazam, sold under the brand names Frisium, Onfi and others, is a benzodiazepine class medication that was patented in 1968.[1] Clobazam was first synthesized in 1966 and first published in 1969. Clobazam was originally marketed as an anxioselective anxiolytic since 1970,[2][3] and an anticonvulsant since 1984.[4] The primary drug-development goal was to provide greater anxiolytic, anti-obsessive efficacy with fewer benzodiazep... [wikipedia]
* May have multiple approval dates, manufacturers, or labelers.1 Discussion
Dosage List
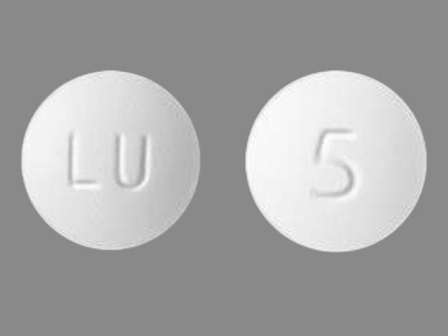
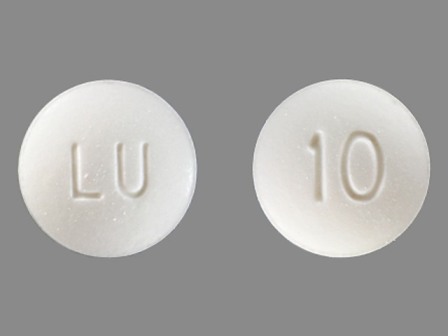
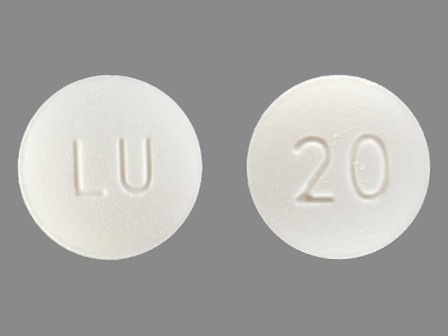
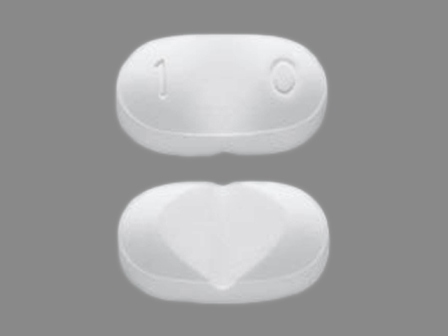

Popular Topics
can onfi cause soreness in hamstrings 2 REPLIES
2 REPLIES
I started onfi and my hamstrings are tight and sore, so is my sacrum area. Is this a bad side effect? ## Onfi contains t...
Baclofen, Lamictal, Keppra, Onfi, Ibuprofen and Flexeril 1 REPLY
1 REPLY
My Dr put me on baclofen for rls and I was also prescribed 600 mg ibuprofen for pain in the hospital. I had fluid around...