Hycamtin
Active Ingredient(s): TopotecanFDA Approved: * May 28, 1996
Pharm Company: * GLAXOSMITHKLINE
Category: Cancer
Topotecan, sold under the brand name Hycamtin among others, is a chemotherapeutic agent medication that is a topoisomerase inhibitor. It is a synthetic, water-soluble analog of the natural chemical compound camptothecin. It is used in the form of its hydrochloride salt to treat ovarian cancer, lung cancer and other cancer types. After GlaxoSmithKline received final FDA approval for topotecan on October 15, 2007, it became the first topoisomerase I inhibitor for oral use.[citation nee... [wikipedia]
* May have multiple approval dates, manufacturers, or labelers.Dosage List
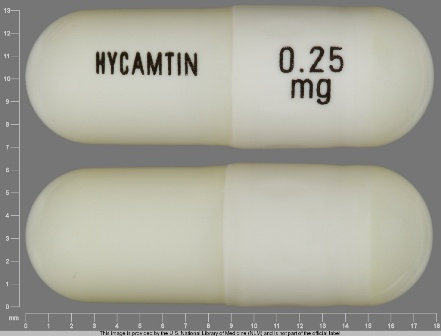
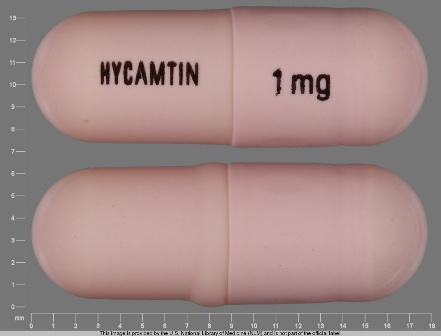



Hycamtin 4 mg/4ml Intravenous Injection, Powder, Lyophilized, for Solution
NDC: 0007-4201
Labeler:
Glaxosmithkline LLC
Hycamtin 4 mg/4ml Intravenous Injection, Powder, Lyophilized, for Solution
NDC: 0078-0674
Labeler:
Novartis Pharmaceuticals Corporation
Hycamtin 4 mg/4ml Intravenous Injection, Powder, Lyophilized, for Solution
NDC: 12064-002
Labeler:
Glaxosmithkline Manufacturing Spa