Cardura
Active Ingredient(s): DoxazosinFDA Approved: * November 2, 1990
Pharm Company: * PFIZER
Category: Blood Pressure
Doxazosin, sold under the brand names Cardura among others, is a medication used to treat symptoms of benign prostatic hyperplasia (enlarged prostate) and hypertension (high blood pressure).[1][2] For high blood pressure, it is a less preferred option.[2] It is taken by mouth.[2] Common side effects include dizziness, sleepiness, swelling, nausea, shortness of breath, and abdominal pain.[2] Severe side effects may... [wikipedia]
* May have multiple approval dates, manufacturers, or labelers.3 Discussions
Dosage List
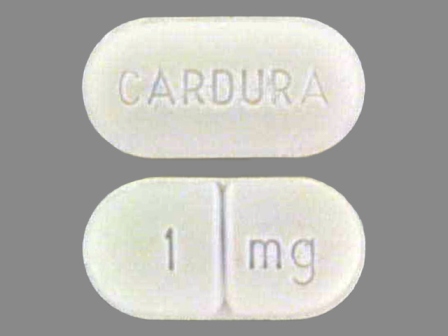

Related Brands
Drugs with the same active ingredientsPopular Topics
Drug Interactions With Cardura 2 REPLIES
2 REPLIES
Is there an adverse reaction between Cardura and SMZ-TMP? ## No, I didn't find any interactions listed between the t...
Interactions between Anadin Extra, Cardura, Lisinopril, Pentasa, Lipitor and Metformin
Can I take Anadin Extra with Cardura, Lisinopril, Pentasa, Lipitor and Metformin? Are there any side effects?...