Afinitor
Active Ingredient(s): EverolimusFDA Approved: * March 30, 2009
Pharm Company: * NOVARTIS PHARMS
Category: Cancer
Everolimus, sold under the brand name Afinitor among others, is a medication used as an immunosuppressant to prevent rejection of organ transplants[8] and as a targeted therapy in the treatment of renal cell cancer and other tumours.[9] It is the 40-O-(2-hydroxyethyl) derivative of sirolimus and works similarly to sirolimus as an inhibitor of mammalian target of rapamycin (mTOR).[10] It is marketed by Novartis under the trade names Zortress (US)... [wikipedia]
* May have multiple approval dates, manufacturers, or labelers.3 Discussions
Dosage List
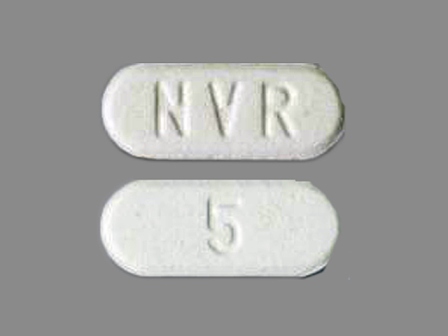
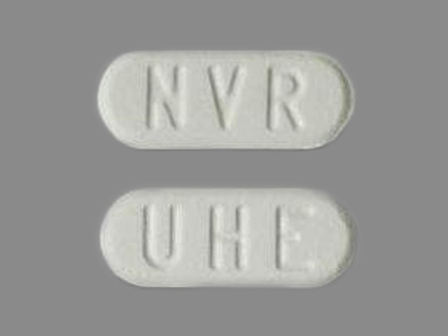
Related Brands
Drugs with the same active ingredientsPopular Topics
Afinitor reviews 1 REPLY
1 REPLY
Can anyone share their experience with taking afinitor, mainly the side effects, especially if you are taking it for neu...
Renal cell carcinoma and Afinitor 1 REPLY
1 REPLY
My daughter has stage 4 renal cell carcinoma, the cancer has metastasized to her lungs, lymph nodes and vena cava vein. ...