70518-2157 : Effexor 75 mg Oral Capsule, Extended Release
| NDC: | 70518-2157 |
| Labeler: | Remedyrepack Inc. |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Effexor XR Effexor XR |
| Dosage Form: | Oral Capsule, Extended Release |
| Application #: | NDA020699 |
| Rev. Date: |
Appearance:
| Markings: | W;EffexorXR;75 |
| Shapes: |
Capsule |
| Colors: |
 Orange Orange |
| Size (mm): | 20 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
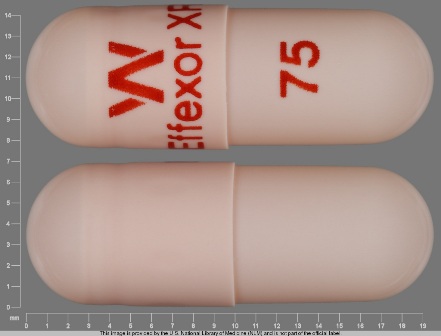
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 70518-2157-0: 30 CAPSULE, EXTENDED RELEASE IN 1 BLISTER PACK (70518‑2157‑0)
Active Ingredients:
- Venlafaxine Hydrochloride
Dosage Strength:
- 75 mg
Inactive Ingredients:
- Powdered Cellulose
- Gelatin, Unspecified
- Ferric Oxide Red
- Titanium Dioxide
- Ethylcellulose (100 Mpa.s)
- Hypromellose, Unspecified
- Hypromellose 2910 (6 Mpa.s) /
Pharmaceutical Classes:
- Norepinephrine Uptake Inhibitors [MoA]
- Serotonin and Norepinephrine Reuptake Inhibitor [EPC]
- Serotonin Uptake Inhibitors [MoA]
Related Products:
Based on records with the same trade name.- 70518-2156 Effexor 150 mg Oral Capsule, Extended Release by Remedyrepack Inc.
- 24236-913 Effexor 75 mg Oral Capsule, Extended Release by Remedyrepack Inc.
- 24236-963 Effexor 150 mg Oral Capsule, Extended Release by Remedyrepack Inc.
- 58151-125 Effexor 37.5 mg Oral Capsule, Extended Release by Viatris Specialty LLC
- 58151-126 Effexor 75 mg Oral Capsule, Extended Release by Viatris Specialty LLC
- 58151-127 Effexor 150 mg Oral Capsule, Extended Release by Viatris Specialty LLC
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 70518-2156Next: 70518-2158 >
Related Discussions:
effexor venlafaxine withdrawals
My daughter has been trying to taper off effexor for 6 months. She has had nausea, vertigo, brain zaps. A couple days ag... 3 replies
My daughter has been trying to taper off effexor for 6 months. She has had nausea, vertigo, brain zaps. A couple days ag... 3 replies
venlafaxine 75mg generic for effexor
i have taken effexof for years and i have never seen any like the one i have now. it is pale yellow round tablet smooth ... 4 replies
i have taken effexof for years and i have never seen any like the one i have now. it is pale yellow round tablet smooth ... 4 replies
Switched from effexor(venlafaxine) to celexa (citalopram)
Hi....Here's the short version of my story. I was on effexor for 13 years, started out at 37.5mg for probably 9 year... 1 reply
Hi....Here's the short version of my story. I was on effexor for 13 years, started out at 37.5mg for probably 9 year... 1 reply
Venlafaxine XR and Dizziness/Vertigo
My doctor changed me from Effexor XR capsules 37.5 mg. to 37.5 mg Venlafaxine XR (white tablet). I am now having a lot o... 7 replies
My doctor changed me from Effexor XR capsules 37.5 mg. to 37.5 mg Venlafaxine XR (white tablet). I am now having a lot o... 7 replies
Venlafaxine XR coverage changes
I was just informed by my pharmacy today today that my insurance will no longer allowe me to take Venlafaxine XR 150mg t... 2 replies
I was just informed by my pharmacy today today that my insurance will no longer allowe me to take Venlafaxine XR 150mg t... 2 replies
VENLAFAXINE HCL 75MG 24HR SA CAP
I have been on Cymbalta for RSD/CRPS pain and my VA doctor wants me to switch to VENLAFAXINE HCL 75MG 24HR SA CAP. She i... 8 replies
I have been on Cymbalta for RSD/CRPS pain and my VA doctor wants me to switch to VENLAFAXINE HCL 75MG 24HR SA CAP. She i... 8 replies
Venlafaxine Hcl 150 mg 24 HR SA Tab
I want to get off of venlafaxine HCL 150 mg 24 HR SA Tab. There is no score line. Can I cut it in half and take just hal... 5 replies
I want to get off of venlafaxine HCL 150 mg 24 HR SA Tab. There is no score line. Can I cut it in half and take just hal... 5 replies
Venlafaxine HCL ER manufactured by Aurobino contact FDA
I have been taking generic Venlafaxine from Tega Pharma for the past 5 years and have had no issues. My pharmacy is now ... 5 replies
I have been taking generic Venlafaxine from Tega Pharma for the past 5 years and have had no issues. My pharmacy is now ... 5 replies
Venlafaxine Er 37 5
Dr give me Venlafaxine ER 37.5 I took 1 pill a day after a week increase to 2 pills a day.. well I finish the medication... 3 replies
Dr give me Venlafaxine ER 37.5 I took 1 pill a day after a week increase to 2 pills a day.. well I finish the medication... 3 replies
Venlafaxine Hcl 37 5 Mg Tablet
my hematology oncologist prescribe this to me for "hot flashes". I had breast cancer and a total hysterectomy an... 2 replies
my hematology oncologist prescribe this to me for "hot flashes". I had breast cancer and a total hysterectomy an... 2 replies
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.