70518-0617 : Metoclopramide 10 mg Oral Tablet
| NDC: | 70518-0617 |
| Labeler: | Remedyrepack Inc. |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Metoclopramide Metoclopramide |
| Dosage Form: | Oral Tablet |
| Application #: | ANDA070581 |
| Rev. Date: |
Appearance:
| Markings: | 685 |
| Shapes: |
Round |
| Colors: |
 White White |
| Size (mm): | 8 |
| Segments: * | 2 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 2 indicates a scored pill which can be broken into 2 equal pieces. | |
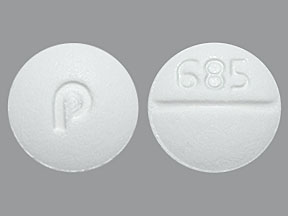
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 70518-0617-0: 30 TABLET IN 1 BLISTER PACK (70518‑0617‑0)
Active Ingredients:
- Metoclopramide Hydrochloride
Dosage Strength:
- 10 mg
Inactive Ingredients:
- Silicon Dioxide
- Starch, Corn
- Magnesium Stearate
- Mannitol
Pharmaceutical Classes:
- Dopamine D2 Antagonists [MoA]
- Dopamine-2 Receptor Antagonist [EPC]
Related Products:
Based on records with the same trade name.- 70518-0669 Metoclopramide 10 mg Oral Tablet by Remedyrepack Inc.
- 70518-1193 Metoclopramide 10 mg Oral Tablet by Remedyrepack Inc.
- 70518-4541 Metoclopramide 10 mg Oral Tablet by Remedyrepack Inc.
- 24236-485 Metoclopramide 10 mg (As Metoclopramide Hydrochloride) Oral Tablet by Remedyrepack Inc.
- 52125-050 Metoclopramide 10 mg (As Metoclopramide Hydrochloride) Oral Tablet by Remedyrepack Inc.
- 52125-122 Metoclopramide 10 mg (As Metoclopramide Hydrochloride) Oral Tablet by Remedyrepack Inc.
- 52125-557 Metoclopramide 10 mg (As Metoclopramide Hydrochloride) Oral Tablet by Remedyrepack Inc.
- 52125-652 Metoclopramide 10 mg (As Metoclopramide Hydrochloride) Oral Tablet by Remedyrepack Inc.
- 0093-2203 Metoclopramide 10 mg (As Metoclopramide Hydrochloride) Oral Tablet by Teva Pharmaceuticals USA Inc
- 0093-2204 Metoclopramide 5 mg (As Metoclopramide Hydrochloride) Oral Tablet by Teva Pharmaceuticals USA Inc
- 0121-0576 Metoclopramide 5 mg/5ml Oral Solution by Pharmaceutical Associates, Inc.
- 0121-1576 Metoclopramide 5 mg/5ml Oral Solution by Pharmaceutical Associates, Inc.
- 0228-2269 Metoclopramide 10 mg (As Metoclopramide Hydrochloride) Oral Tablet by Actavis Elizabeth LLC
- 0404-9910 Metoclopramide 5 mg/ml Intramuscular; Intravenous Injection, Solution by Henry Schein, Inc.
- 0409-3414 Metoclopramide 5 mg/ml Intramuscular; Intravenous Injection, Solution by Hospira, Inc.
- 0409-5255 Metoclopramide 5 mg/ml Intramuscular; Intravenous Injection, Solution by Hospira, Inc.
- 0440-1771 Metoclopramide 10 mg (As Metoclopramide Hydrochloride) Oral Tablet by Liberty Pharmaceuticals, Inc.
- 0603-4614 Metoclopramide 5 mg (As Metoclopramide Hydrochloride) Oral Tablet by Qualitest Pharmaceuticals
- 0603-4615 Metoclopramide 10 mg (As Metoclopramide Hydrochloride) Oral Tablet by Qualitest Pharmaceuticals
- 0615-2536 Metoclopramide 10 mg (As Metoclopramide Hydrochloride) Oral Tablet by Ncs Healthcare of Ky, Inc Dba Vangard Labs
- More related products ...
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 70518-0616Next: 70518-0618 >
Related Discussions:
Metoclopramide for Migraine Headaches
I have a Migraine Headache & my Dr prescribed this. Will it work? And will it make me sleepy? ## Metoclopramide is a... 2 replies
I have a Migraine Headache & my Dr prescribed this. Will it work? And will it make me sleepy? ## Metoclopramide is a... 2 replies
Metoclopramide used for?
What is Metoclopramide used for? ## Hello, Metoclopramide HCl is primarily used to treat nausea and vomiting. For more i... 1 reply
What is Metoclopramide used for? ## Hello, Metoclopramide HCl is primarily used to treat nausea and vomiting. For more i... 1 reply
what is bio metoclopramide 10 used for
I've been given these pills in the hospital and they didn't explain what this is for. Can you please help me out... 1 reply
I've been given these pills in the hospital and they didn't explain what this is for. Can you please help me out... 1 reply
generic name for metoclopramide
I have a oblong white pill in my medicine bottle that has a NO23 on it. I wanted to know what was a generic medicine nam... 2 replies
I have a oblong white pill in my medicine bottle that has a NO23 on it. I wanted to know what was a generic medicine nam... 2 replies
metoclopramide 5mg/5ml syrup
reflux...
reflux...
metoclopramide side affects
reflux...
reflux...
metoclopramide 10mg
take 1 tablet before meals...
take 1 tablet before meals...
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.