57844-009 : Adipex-p 37.5 mg Oral Tablet
| NDC: | 57844-009 |
| Labeler: | Teva Select Brands |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Adipex-p Adipex-p |
| Dosage Form: | Oral Tablet |
| Application #: | ANDA085128 |
| Rev. Date: | |
| CSA Schedule: | CIV (US) [1] |
[1] Schedule IV Controlled Substance: Low potential for abuse relative to substances in Schedule III. Examples include Alprazolam (Xanax), Diazepam (Valium), Carisoprodol (Soma), Clonazepam (Klonopin), Lorazepam (Ativan), Clorazepate (Tranxene), Midazolam (Versed), Temazepam (Restoril), and Triazolam (Halcion).. More Details: US Dept of Justice Controlled Substance Schedules.
Appearance:
| Markings: | ADIPEX;P;9;9 |
| Shapes: |
Oval |
| Colors: |
 Blue / Blue /
 White White |
| Size (mm): | 10 |
| Segments: * | 2 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 2 indicates a scored pill which can be broken into 2 equal pieces. | |
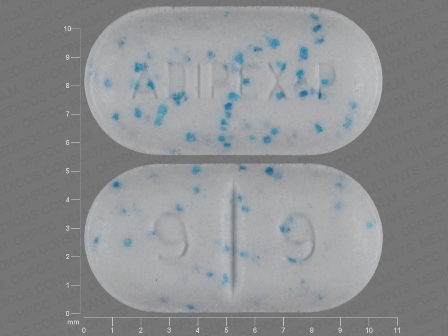
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 57844-009-00: 40 TABLET IN 1 BOTTLE (57844‑009‑00)
- 57844-009-01: 100 TABLET IN 1 BOTTLE (57844‑009‑01)
- 57844-009-10: 1000 TABLET IN 1 BOTTLE (57844‑009‑10)
- 57844-009-56: 30 TABLET IN 1 BOTTLE (57844‑009‑56)
Active Ingredients:
- Phentermine Hydrochloride
Dosage Strength:
- 37.5 mg
Inactive Ingredients:
- Starch, Corn
- Anhydrous Lactose
- Magnesium Stearate
- Cellulose, Microcrystalline
- Sucrose
- Fd&c Blue No. 1
Pharmaceutical Classes:
- Appetite Suppression [PE]
- Increased Sympathetic Activity [PE]
- Sympathomimetic Amine Anorectic [EPC]
Related Products:
Based on records with the same trade name.- 57844-019 Adipex-p 37.5 mg Oral Capsule by Teva Select Brands
- 57844-140 Adipex-p 37.5 mg Oral Tablet by Teva Pharmaceuticals USA, Inc.
- 50090-0284 Adipex-p 37.5 mg Oral Tablet by A-s Medication Solutions LLC
- 54569-1718 Adipex-p 37.5 mg Oral Tablet by A-s Medication Solutions LLC
- 54868-0479 Adipex-p 37.5 mg Oral Tablet by Physicians Total Care, Inc.
- 55289-379 Adipex-p 37.5 mg Oral Tablet by Pd-rx Pharmaceuticals, Inc.
- 72789-175 Adipex-p 37.5 mg Oral Tablet by Pd-rx Pharmaceuticals, Inc.
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 57842-7734Next: 57844-019 >
Related Discussions:
Phentermine vs Adipex MP237
I was prescribed phentermine 37.5mg and I got the Adipex pill that has blue specs with MP237 on it. It had very little e... 1 reply
I was prescribed phentermine 37.5mg and I got the Adipex pill that has blue specs with MP237 on it. It had very little e... 1 reply
Phentermine anyone have strange side effects?
I just started taking this medicine for weight loss. I am having hot flashes and cold chills, terrible headache and a ti... 356 replies
I just started taking this medicine for weight loss. I am having hot flashes and cold chills, terrible headache and a ti... 356 replies
Phentermine side effects
What are the side effects of phentermine? ## Side effects are dry mouth, restlessness, anxiety, all in all not to bad co... 75 replies
What are the side effects of phentermine? ## Side effects are dry mouth, restlessness, anxiety, all in all not to bad co... 75 replies
phentermine imprint code k25
K 25 IS IMPRINTED ON THE PILL AND IT IS WHITE AND BLUE SPECKELED OVAL SHAPED. ## This is an over the counter product sol... 41 replies
K 25 IS IMPRINTED ON THE PILL AND IT IS WHITE AND BLUE SPECKELED OVAL SHAPED. ## This is an over the counter product sol... 41 replies
Phentermine 37.5 mg with A 159
Qualis Pharmaceutical white/blue speckled Phentermine 37.5 mg with A 159 stamped on it...are these real? ## Phentermine ... 19 replies
Qualis Pharmaceutical white/blue speckled Phentermine 37.5 mg with A 159 stamped on it...are these real? ## Phentermine ... 19 replies
Phentermine Bend, Oregon
I need to find a doctor in Bend, Oregon (or even nearby) that prescribes Phentermine. NOT phentermine-d. ## Could someon... 19 replies
I need to find a doctor in Bend, Oregon (or even nearby) that prescribes Phentermine. NOT phentermine-d. ## Could someon... 19 replies
Phentermine Prescribers
I was taking Phentermine 37.5 mg for a couple months and lost about 20lbs. The doctor I was seeing is no longer prescrib... 8 replies
I was taking Phentermine 37.5 mg for a couple months and lost about 20lbs. The doctor I was seeing is no longer prescrib... 8 replies
Phentermine and back pain
Hi everyone. I'm new to phentermine (today will be the 4th half of a 37.5 MG pill) and I must say it's giving me... 7 replies
Hi everyone. I'm new to phentermine (today will be the 4th half of a 37.5 MG pill) and I must say it's giving me... 7 replies
Phentermine not working
I have taken phentermine several times and always had great results. I was prescribed phentermine 37.5 mg a few weeks ag... 6 replies
I have taken phentermine several times and always had great results. I was prescribed phentermine 37.5 mg a few weeks ag... 6 replies
phentermine k 25 export quality
Why would a bubble pack of phentermine k-25 have export quality written on the back ## I got the same thing from India. ... 6 replies
Why would a bubble pack of phentermine k-25 have export quality written on the back ## I got the same thing from India. ... 6 replies
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.