54868-5231 : Emend 80 mg Oral Capsule
| NDC: | 54868-5231 |
| Labeler: | Physicians Total Care, Inc. |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Emend Emend |
| Dosage Form: | Oral Capsule |
| Application #: | NDA021549 |
| Rev. Date: |
Appearance:
| Markings: | 461;80;mg |
| Shapes: |
Capsule |
| Colors: |
 White White |
| Size (mm): | 17 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
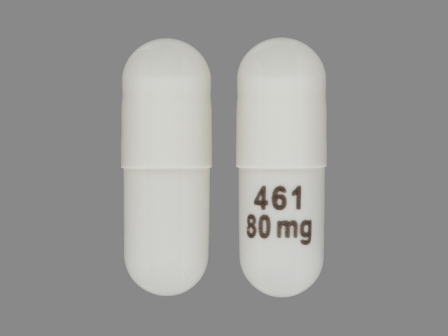
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 54868-5231-1: 1 BLISTER PACK IN 1 CARTON (54868‑5231‑1) > 6 CAPSULE IN 1 BLISTER PACK
- 54868-5231-2: 2 CAPSULE IN 1 DOSE PACK (54868‑5231‑2)
- 54868-5231-3: 4 CAPSULE IN 1 DOSE PACK (54868‑5231‑3)
Active Ingredients:
- Aprepitant
Dosage Strength:
- 80 mg
Inactive Ingredients:
- Sucrose
- Cellulose, Microcrystalline
- Hydroxypropyl Cellulose
- Sodium Lauryl Sulfate
- Gelatin
- Titanium Dioxide
- Silicon Dioxide
Pharmaceutical Classes:
- Neurokinin 1 Antagonists [MoA]
- Substance P/Neurokinin-1 Receptor Antagonist [EPC]
- Cytochrome P450 3A4 Inhibitors [MoA]
- Cytochrome P450 2C9 Inducers [MoA]
- Cytochrome P450 3A4 Inducers [MoA]
Related Products:
Based on records with the same trade name.- 54868-5325 Emend Tri-fold Pack by Physicians Total Care, Inc.
- 0006-0461 Emend 80 mg Oral Capsule by Merck Sharp & Dohme Corp.
- 0006-0462 Emend 125 mg Oral Capsule by Merck Sharp & Dohme Corp.
- 0006-0464 Emend 40 mg Oral Capsule by Merck Sharp & Dohme Corp.
- 0006-3061 Emend 150 mg/5ml Intravenous Injection, Powder, Lyophilized, for Solution by Merck Sharp & Dohme Corp.
- 0006-3066 Emend 125 mg Oral Powder, for Suspension by Merck Sharp & Dohme Corp.
- 0006-3862 Emend Tri-fold Pack by Merck Sharp & Dohme Corp.
- 0006-3884 Emend 115 mg/5ml Intravenous Injection, Powder, Lyophilized, for Solution by Merck Sharp & Dohme Corp.
- 0006-3941 Emend 150 mg/5ml Intravenous Injection, Powder, Lyophilized, for Solution by Merck Sharp & Dohme Corp.
- 42254-160 Emend 40 mg Oral Capsule by Rebel Distributors Corp
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 54868-5230Next: 54868-5232 >
Related Discussions:
Pantoprazole 40mg and Aprepitant 80
I am taking Pantoprazole for severe acid reflux. Yesterday I started chemotherapy for breast cancer and have been prescr... 1 reply
I am taking Pantoprazole for severe acid reflux. Yesterday I started chemotherapy for breast cancer and have been prescr... 1 reply
Are people taking Emend who do not have cancer?
Hi, I have severe gastroparesis for 5 years now. I’ve been through tubes, pics, ports, pyloroplasty and Botox. I a... 1 reply
Hi, I have severe gastroparesis for 5 years now. I’ve been through tubes, pics, ports, pyloroplasty and Botox. I a... 1 reply
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.