53014-726 : Dipentum 250 mg Oral Capsule
| NDC: | 53014-726 |
| Labeler: | Ucb, Inc. |
| Drug Name: |  Dipentum Dipentum |
| Rev. Date: |
Appearance:
| Markings: | DIPENTUM;250;mg |
| Shapes: |
Capsule |
| Colors: |
 White White |
| Size (mm): | 19 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
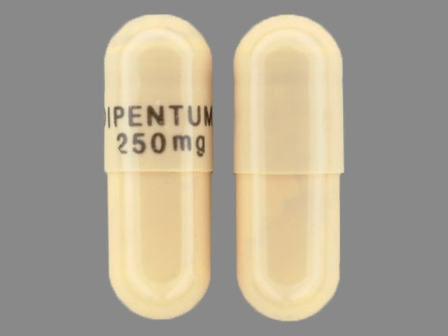
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
Active Ingredients:
- Olsalazine
Inactive Ingredients:
- Magnesium Stearate
- Black Iron Oxide
- Caramel
- Gelatin
- Titanium Dioxide
Related Products:
Based on records with the same trade name.- 0037-6860 Dipentum 250 mg Oral Capsule, Gelatin Coated by Meda Pharmaceuticals
- 68151-0312 Dipentum 250 mg Oral Capsule, Gelatin Coated by Carilion Materials Management
- 68151-3120 Dipentum 250 mg Oral Capsule, Gelatin Coated by Carilion Materials Management
- 68220-160 Dipentum 250 mg Oral Capsule by Alaven Pharmaceutical LLC
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 53014-594Next: 53014-847 >
Related Discussions:
dipentum, cost
Each year for the last several my co-pay for dipentum has increased dramatically, now to $250 per month from about $40. ... 2 replies
Each year for the last several my co-pay for dipentum has increased dramatically, now to $250 per month from about $40. ... 2 replies
dipentum verses sufasalazine
lost job can no longer aford high cost of dipentum for my chrons disease am wondering if sufasalazine will do as well as...
lost job can no longer aford high cost of dipentum for my chrons disease am wondering if sufasalazine will do as well as...
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.