35573-417 : Atomoxetine 25 mg Oral Capsule
| NDC: | 35573-417 |
| Labeler: | Burel Pharma |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Atomoxetine Atomoxetine |
| Dosage Form: | Oral Capsule |
| Application #: | ANDA079016 |
| Rev. Date: |
Appearance:
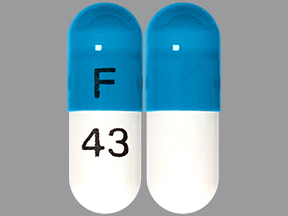
| Markings: | F;43 |
| Shapes: |
Capsule |
| Colors: |
 Blue / Blue /
 White White |
| Size (mm): | 14 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 35573-417-30: 30 CAPSULE IN 1 BOTTLE (35573‑417‑30)
Active Ingredients:
- Atomoxetine Hydrochloride
Dosage Strength:
- 25 mg
Inactive Ingredients:
- Starch, Corn
- Dimethicone
- Silicon Dioxide
- Gelatin, Unspecified
- Titanium Dioxide
- Sodium Lauryl Sulfate
- Fd&c Blue No. 2
- Ferrosoferric Oxide
- Shellac /
Pharmaceutical Classes:
- Norepinephrine Reuptake Inhibitor [EPC]
- Norepinephrine Uptake Inhibitors [MoA]
Related Products:
Based on records with the same trade name.- 35573-415 Atomoxetine 10 mg Oral Capsule by Burel Pharma
- 35573-416 Atomoxetine 18 mg Oral Capsule by Burel Pharma
- 35573-418 Atomoxetine 40 mg Oral Capsule by Burel Pharma
- 35573-419 Atomoxetine 60 mg Oral Capsule by Burel Pharma
- 35573-420 Atomoxetine 80 mg Oral Capsule by Burel Pharma
- 35573-421 Atomoxetine 100 mg Oral Capsule by Burel Pharma
- 0093-3542 Atomoxetine 10 mg Oral Capsule by Teva Pharmaceuticals USA, Inc.
- 0093-3543 Atomoxetine 18 mg Oral Capsule by Teva Pharmaceuticals USA, Inc.
- 0093-3544 Atomoxetine 25 mg Oral Capsule by Teva Pharmaceuticals USA, Inc.
- 0093-3545 Atomoxetine 40 mg Oral Capsule by Teva Pharmaceuticals USA, Inc.
- 0093-3546 Atomoxetine 60 mg Oral Capsule by Teva Pharmaceuticals USA, Inc.
- 0093-3547 Atomoxetine 80 mg Oral Capsule by Teva Pharmaceuticals USA, Inc.
- 0093-3548 Atomoxetine 100 mg Oral Capsule by Teva Pharmaceuticals USA, Inc.
- 0904-6908 Atomoxetine 40 mg Oral Capsule by Major Pharmaceuticals
- 16714-755 Atomoxetine 10 mg Oral Capsule by Northstar Rx LLC
- 16714-756 Atomoxetine 18 mg Oral Capsule by Northstar Rx LLC
- 16714-757 Atomoxetine 25 mg Oral Capsule by Northstar Rx LLC
- 16714-758 Atomoxetine 40 mg Oral Capsule by Northstar Rx LLC
- 16714-759 Atomoxetine 60 mg Oral Capsule by Northstar Rx LLC
- 16714-760 Atomoxetine 80 mg Oral Capsule by Northstar Rx LLC
- More related products ...
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 35573-416Next: 35573-418 >
Related Discussions:
Strattera (Atomoxetine) - Good or Bad Generic Brands
Do Atomoxetine generics have good or bad brands, and if so, which ones? Anyone who has been on an Adderall forum knows h...
Do Atomoxetine generics have good or bad brands, and if so, which ones? Anyone who has been on an Adderall forum knows h...
Atomoxetine drug information
-Drug details/uses -Side effects -Pharmacology -Half-life -Misc...
-Drug details/uses -Side effects -Pharmacology -Half-life -Misc...
When Atomoxetine Hydrochloride Capsules Are Used As Medicine
in which disease Atomoxetine hydrochloride capsules are used?...
in which disease Atomoxetine hydrochloride capsules are used?...
Quelbree (Viloxazine) vs Strattera (Atomoxetine)
Quelbree (Viloxazine) vs Strattera (Atomoxetine) Has anyone tried both who can compare them? ## I spelled Qelbree wrong.... 1 reply
Quelbree (Viloxazine) vs Strattera (Atomoxetine) Has anyone tried both who can compare them? ## I spelled Qelbree wrong.... 1 reply
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.