0430-0470 : Actonel 30 mg Oral Tablet
| NDC: | 0430-0470 |
| Labeler: | Warner Chilcott (Us), LLC |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Actonel Actonel |
| Dosage Form: | Oral Tablet, Film Coated |
| Application #: | NDA020835 |
| Rev. Date: |
Appearance:
| Markings: | RSN;30;MG |
| Shapes: |
Oval |
| Colors: |
 White White |
| Size (mm): | 12 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
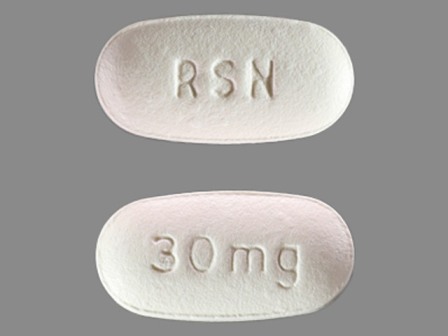
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 0430-0470-15: 30 TABLET, FILM COATED IN 1 BOTTLE (0430‑0470‑15)
Active Ingredients:
- Risedronate Sodium Hemi-pentahydrate
- Risedronate Sodium Monohydrate
Dosage Strength:
- 25.8 mg
- 4.2 mg
Inactive Ingredients:
- Crospovidone
- Hydroxypropyl Cellulose (Type H)
- Hypromelloses
- Magnesium Stearate
- Cellulose, Microcrystalline
- Polyethylene Glycols
- Silicon Dioxide
- Titanium Dioxide
- Lactose Monohydrate
Pharmaceutical Classes:
- Bisphosphonate [EPC]
- Diphosphonates [Chemical/Ingredient]
Related Products:
Based on records with the same trade name.- 0430-0471 Actonel 5 mg Oral Tablet by Warner Chilcott (Us), LLC
- 0430-0472 Actonel 35 4-week Once Weekly Dosepack by Warner Chilcott (Us), LLC
- 0430-0477 Actonel 75 Dosepack by Warner Chilcott (Us), LLC
- 0430-0478 Actonel 150 mg Oral Tablet by Warner Chilcott (Us), LLC
- 0149-0470 Actonel 30 mg Oral Tablet by Warner Chilcott Pharmaceuticals Inc.
- 0149-0471 Actonel 5 mg Oral Tablet by Warner Chilcott Pharmaceuticals Inc.
- 0149-0472 Actonel 35 4-week Once Weekly Dosepack by Warner Chilcott Pharmaceuticals Inc.
- 0149-0477 Actonel 75 Dosepack by Warner Chilcott Pharmaceuticals Inc.
- 0149-0478 Actonel 150 mg Oral Tablet by Warner Chilcott Pharmaceuticals Inc.
- 54868-4386 Actonel 5 mg Oral Tablet by Physicians Total Care, Inc.
- 54868-4671 Actonel 35 4-week Once Weekly Dosepack by Physicians Total Care, Inc.
- 54868-6069 Actonel 150 mg Oral Tablet by Physicians Total Care, Inc.
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 0430-0420Next: 0430-0471 >
Related Discussions:
Is morphine sulfate pentahydrate synthetic?
I have been taking morphine sulfate (Statex) and codeine for 20 years without any problems at all. I am extremely intole... 3 replies
I have been taking morphine sulfate (Statex) and codeine for 20 years without any problems at all. I am extremely intole... 3 replies
Actonel Side Effects
Has anyone taking Actonel 35 mg experienced severe joint pain? ## have you experience hand shakes? ## Actonel Side Effec... 4 replies
Has anyone taking Actonel 35 mg experienced severe joint pain? ## have you experience hand shakes? ## Actonel Side Effec... 4 replies
Sodium Valproate and pregnancy
I have epilepsy and have taken Sodium Valproate 300 mg daily for nearly 5 years. I am now pregnant and want to know if t... 7 replies
I have epilepsy and have taken Sodium Valproate 300 mg daily for nearly 5 years. I am now pregnant and want to know if t... 7 replies
Sodium level coming down
My mother is 79yrs old. She has had an Electrolyte problem for the last 2 yrs. Sodium levels are coming down. This is th... 4 replies
My mother is 79yrs old. She has had an Electrolyte problem for the last 2 yrs. Sodium levels are coming down. This is th... 4 replies
Sodium Hyaluronate or Hyaluronic Acid with Warfarin
Can Sodium Hyaluronate or Hyaluronic Acid be taken with Warfarin? ## Sorry, I am not finding any information on this. Ha... 2 replies
Can Sodium Hyaluronate or Hyaluronic Acid be taken with Warfarin? ## Sorry, I am not finding any information on this. Ha... 2 replies
sodium lauryl sulfate and sodium laureth sulfate
how is this helpful in medicine - like bystolic 5mg for high blood pressure? I thought it was a foaming agent for deterg... 2 replies
how is this helpful in medicine - like bystolic 5mg for high blood pressure? I thought it was a foaming agent for deterg... 2 replies
Sodium Tetraborate Information and Safety
What are the dangers of Sodium Tetraborate? Is it harmful if swallowed by a toddler of 11 months? Do I need to call pois... 1 reply
What are the dangers of Sodium Tetraborate? Is it harmful if swallowed by a toddler of 11 months? Do I need to call pois... 1 reply
Sodium level stays at 135
My father's (58 yrs old) sodium level stays at 135 and he is taking natrise 15 mg on alternative days but still he s... 1 reply
My father's (58 yrs old) sodium level stays at 135 and he is taking natrise 15 mg on alternative days but still he s... 1 reply
Sodium Valproate Ingridients
i"m looking for the ingredients of sodium val as my nephew has angel man syndrome and has been diagnosed with epilep... 1 reply
i"m looking for the ingredients of sodium val as my nephew has angel man syndrome and has been diagnosed with epilep... 1 reply
sodium chloride pills
why are they hard on your stomach ## Sodium Chloride is basically just salt, and while all medications are hard on your ... 1 reply
why are they hard on your stomach ## Sodium Chloride is basically just salt, and while all medications are hard on your ... 1 reply
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.