0378-2005 : Paroxetine (As Paroxetine Hydrochloride) 37.5 mg Extended Release Tablet
| NDC: | 0378-2005 |
| Labeler: | Mylan Pharmaceuticals Inc. |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Paroxetine Hydrochloride Paroxetine Hydrochloride |
| Dosage Form: | Oral Tablet, Film Coated, Extended Release |
| Application #: | ANDA091427 |
| Rev. Date: |
Appearance:
| Markings: | M;P5 |
| Shapes: |
Round |
| Colors: |
 Orange Orange |
| Size (mm): | 8 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
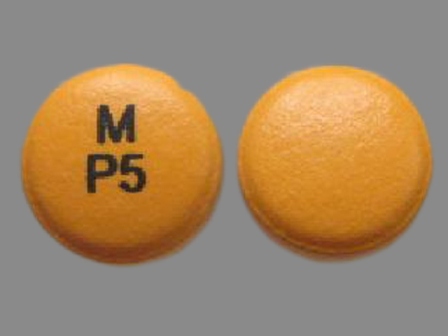
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 0378-2005-01: 100 TABLET, FILM COATED, EXTENDED RELEASE IN 1 BOTTLE (0378‑2005‑01)
- 0378-2005-05: 500 TABLET, FILM COATED, EXTENDED RELEASE IN 1 BOTTLE (0378‑2005‑05)
- 0378-2005-93: 30 TABLET, FILM COATED, EXTENDED RELEASE IN 1 BOTTLE (0378‑2005‑93)
Active Ingredients:
- Paroxetine Hydrochloride Hemihydrate
Dosage Strength:
- 37.5 mg
Inactive Ingredients:
- Ferrosoferric Oxide
- Silicon Dioxide
- D&c Red No. 30
- Fd&c Yellow No. 6
- Hydroxypropyl Cellulose (Type H)
- Hypromelloses
- Lactose Monohydrate
- Lecithin, Soybean
- Magnesium Stearate
- Methacrylic Acid - Ethyl Acrylate Copolymer (1:1) Type a
- Cellulose, Microcrystalline
- Polydextrose
- Polyethylene Glycols
- Polysorbate 80
- Propylene Glycol
- Sodium Alginate
- Sodium Hydroxide
- Talc
- Titanium Dioxide
- Triacetin
- Triethyl Citrate
Pharmaceutical Classes:
- Serotonin Reuptake Inhibitor [EPC]
- Serotonin Uptake Inhibitors [MoA]
Related Products:
Based on records with the same trade name.- 0378-2003 Paroxetine (As Paroxetine Hydrochloride) 12.5 mg Extended Release Tablet by Mylan Pharmaceuticals Inc.
- 0378-2004 Paroxetine (As Paroxetine Hydrochloride) 25 mg Extended Release Tablet by Mylan Pharmaceuticals Inc.
- 42291-579 Paroxetine Hydrochloride 12.5 mg Oral Tablet, Film Coated, Extended Release by Avkare, Inc.
- 42291-580 Paroxetine Hydrochloride 25 mg Oral Tablet, Film Coated, Extended Release by Avkare, Inc.
- 42291-581 Paroxetine Hydrochloride 37.5 mg Oral Tablet, Film Coated, Extended Release by Avkare, Inc.
- 49349-255 Paroxetine 20 mg (As Paroxetine Hydrochloride 22.76 mg ) Oral Tablet by Remedyrepack Inc.
- 49349-285 Paroxetine 40 mg (As Paroxetine Hydrochloride 44.44 mg) Oral Tablet by Remedyrepack Inc.
- 49349-305 Paroxetine 30 mg (As Paroxetine Hydrochloride 34.14 mg) Oral Tablet by Remedyrepack Inc.
- 49349-308 Paroxetine 10 mg (As Paroxetine Hydrochloride 11.38 mg) Oral Tablet by Remedyrepack Inc.
- 49349-364 Paroxetine 20 mg (As Paroxetine Hydrochloride 22.76 mg ) Oral Tablet by Remedyrepack Inc.
- 49349-407 Paroxetine 20 mg (As Paroxetine Hydrochloride 22.76 mg ) Oral Tablet by Remedyrepack Inc.
- 49349-610 Paroxetine 10 mg (As Paroxetine Hydrochloride 11.38 mg) Oral Tablet by Remedyrepack Inc.
- 51079-824 Paroxetine (As Paroxetine Hydrochloride) 12.5 mg Extended Release Tablet by Mylan Institutional Inc.
- 51079-825 Paroxetine (As Paroxetine Hydrochloride) 25 mg Extended Release Tablet by Mylan Institutional Inc.
- 54868-1819 Paroxetine (As Paroxetine Hydrochloride) 25 mg Extended Release Tablet by Physicians Total Care, Inc.
- 54868-6178 Paroxetine (As Paroxetine Hydrochloride) 12.5 mg Extended Release Tablet by Physicians Total Care, Inc.
- 58517-160 Paroxetine 20 mg (As Paroxetine Hydrochloride 22.76 mg ) Oral Tablet by New Horizon Rx Group, LLC
- 60505-1316 Paroxetine Hydrochloride 12.5 mg Oral Tablet, Film Coated, Extended Release by Apotex Corp
- 60505-1317 Paroxetine Hydrochloride 25 mg Oral Tablet, Film Coated, Extended Release by Apotex Corp
- 60505-1318 Paroxetine Hydrochloride 37.5 mg Oral Tablet, Film Coated, Extended Release by Apotex Corp
- More related products ...
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 0378-2004Next: 0378-2008 >
Related Discussions:
Paroxetine manufacturer change
My pharmacy has recently changed the manufacturer from whom they order generic Paroxetine. Are they ANY side effects a p... 8 replies
My pharmacy has recently changed the manufacturer from whom they order generic Paroxetine. Are they ANY side effects a p... 8 replies
Paroxetine Hcl 30mg tab
Markings: 93 7116 blue round film coated ## were or whom makes this generic form of paxil ## oops i mean who makes the b... 3 replies
Markings: 93 7116 blue round film coated ## were or whom makes this generic form of paxil ## oops i mean who makes the b... 3 replies
Paroxetine pill look like?
What should this pill look like? ( 20mg) ## What should Paroxetine 10 mg look like? ## Well that is very hard to say. If... 3 replies
What should this pill look like? ( 20mg) ## What should Paroxetine 10 mg look like? ## Well that is very hard to say. If... 3 replies
paroxetine-higher dosage
i am currently taking paroxetine hcl-40 mg tablet. i had to switch from paxil cr-around 62.5 mg because a program called... 2 replies
i am currently taking paroxetine hcl-40 mg tablet. i had to switch from paxil cr-around 62.5 mg because a program called... 2 replies
Paroxetine 40mg
Round White tablet with imprint ZC18 ## Based on the description provided, I found your pill to be Paroxetine HCL (40 mg... 2 replies
Round White tablet with imprint ZC18 ## Based on the description provided, I found your pill to be Paroxetine HCL (40 mg... 2 replies
paroxetine generic
Round White No Score Imprint ZC18 prescription label has QCPUAGL QCPST100 AUT 191 S0040236869 ## Have never been sent th... 2 replies
Round White No Score Imprint ZC18 prescription label has QCPUAGL QCPST100 AUT 191 S0040236869 ## Have never been sent th... 2 replies
Paroxetine 40 mg pill number zc18
This pill was given to me at giant eagle for Paxil never had high blood pressure until taking. First day got bad headach... 1 reply
This pill was given to me at giant eagle for Paxil never had high blood pressure until taking. First day got bad headach... 1 reply
Paroxetine and body fat help
Hi, I had been on paroxetine 20 mg for about a month, and suddenly have like, a layer of fat on my belly that hasn't... 1 reply
Hi, I had been on paroxetine 20 mg for about a month, and suddenly have like, a layer of fat on my belly that hasn't... 1 reply
PaRoxetineHCI 20 Mg Tablets
I AM TRYING TO FIND OUT ABOUT THIS MED THE DR JUST PUT ME ON AND IF THERE ARE ANY SIDE AFFECTS TO IT AND WHAT THEY ARE T... 1 reply
I AM TRYING TO FIND OUT ABOUT THIS MED THE DR JUST PUT ME ON AND IF THERE ARE ANY SIDE AFFECTS TO IT AND WHAT THEY ARE T... 1 reply
paroxetine hcl 10 mg
The Dr. prescribed me paroxetine hcl 5 weeks ago- for depression- going thru some tough times right now-I took 10 mg for... 1 reply
The Dr. prescribed me paroxetine hcl 5 weeks ago- for depression- going thru some tough times right now-I took 10 mg for... 1 reply
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.